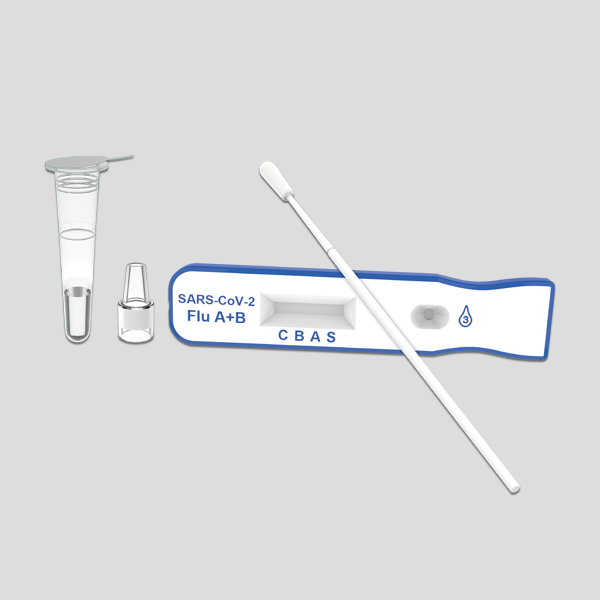
EZER Flu&COVID-19 Antigen Combo Rapid Test
Introduction
Influenza virus belongs to the family of Orthomyxoviridae, and immunologically diverse, single-stranded RNA viruses. There influenza A and B virus is the main pathogen that severe illnesses both in human and in many animal species. Based on the current epidemiological investigation, the incubation period is 1 to 4 days. The main manifestations include acute fever, general aching and respiratory symptoms. Both Type A and B viruses can circulate simultaneously, but usually one type is dominant during a given season.
COVID-19 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection; asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main manifestations include fever, fatigue and dry cough. Nasal congestion, runny nose, sore throat, myalgia and diarrhea are found in a few cases.
Clinical signs and symptoms of respiratory viral infection due to SARS-CoV-2 and influenza can be similar. SARS-CoV-2, influenza A and influenza B viral antigens are generally detectable in upper respiratory specimens during the acute phase of infection.
The EZERTM Flu&COVID-19 Antigen Combo Rapid Test includes influenza antigen rapid test and COVID-19 antigen rapid test, is an immunochromatographic assay for the qualitative detection of 2019 Novel Coronavirus, influenza A and B antigens. The EZERTM Flu&COVID-19 Antigen Combo Rapid Test has four letters on the surface of the strips indicating test line (S)﹑ (A)﹑ (B) and control line (C).
Detection
EZERTM Flu&COVID-19 Antigen Combo Rapid Test is intended for the simultaneous qualitative detection and differentiation of the nucleocapsid protein antigens from SARS-CoV-2, influenza A and influenza B in direct nasal specimens.
Specimen
Nasal
Limit of Detection (LoD)
Flu&COVID-19 :140 TCID50/mL
The minimum detection limit of Flu A for the EZERTM Flu&COVID-19 Antigen Combo Rapid Test was established based on a total of 8 influenza A.
|
Influenza Viral Strain |
Calculated LoD |
|
A/New Caledonia/20/1999_H1N1 |
8.50x103 |
|
A/California/04/2009_H1N1 |
2.11x103 |
|
A/PR/8/34_H1N1 |
2.93x103 |
|
A/Bean Goose/Hubei/chenhu XVI35-1/2016_H3N2 |
4.94x102 |
|
A/Guizhou/54/89_H3N2 |
3.95x102 |
|
A/Human/Hubei/3/2005_H3N2 |
2.93x104 |
|
A/Bar-headed Goose/QH/BTY2/2015_H5N1 |
1.98x105 |
|
A/Anhui/1/2013_H7N9 |
7.90x105 |
The Flu B minimum detection limit for the EZERTM Flu&COVID-19 Antigen Combo Rapid Test was established based on a total of 2 influenza B.
|
Influenza Viral Strain |
Calculated LoD |
|
B/Victoria |
4.25x103 |
|
B/Yamagata |
1.58x102 |
Accuracy
|
|
Influenza A |
Influenza B |
COVID-19 |
|
Relative Sensitivity |
86.8% |
91.7% |
96.6% |
|
Relative Specificity |
94.0% |
97.5% |
100% |
|
Accuracy |
92.2% |
96.1% |
98.9% |
Time to results
Read results at 15minutes and no more than 30 minutes.
Kit storage conditions
2~30°C
Contents
|
Description |
Qty |
|
Test devices |
20 |
|
Sterilized swabs |
20 |
|
Extraction tubes |
20 |
|
Nozzles |
20 |
|
Tube stand |
1 |
|
Package insert |
1 |
Ordering Information
| Product | Cat.No. | Contents |
| EZERTM Flu&COVID-19 Antigen Combo | P213110 | 20 Tests |