China Gold Supplier for Antigen Diagnostic Test - KaiBiLi Flu&Covid-19 Antigen Duo Rapid Test – Genesis
China Gold Supplier for Antigen Diagnostic Test - KaiBiLi Flu&Covid-19 Antigen Duo Rapid Test – Genesis Detail:
Introduction
Clinical signs and symptoms of respiratory viral infection due to SARS-CoV-2 and influenza can be similar. SARS-CoV-2, influenza A and influenza B viral antigens are generally detectable in upper respiratory specimens during the acute phase of infection.
Influenza virus belongs to the family of Orthomyxoviridae, and immunologically diverse, single-stranded RNA viruses. There influenza A and B virus is the main pathogen that severe illnesses both in human and in many animal species. Based on the current epidemiological investigation, the incubation period is 1 to 4 days. The main manifestations include acute fever, general aching and respiratory symptoms. Both Type A and B viruses can circulate simultaneously, but usually one type is dominant during a given season.1
COVID-19 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection; asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main manifestations include fever, fatigue and dry cough. Nasal congestion, runny nose, sore throat, myalgia and diarrhea are found in a few cases.
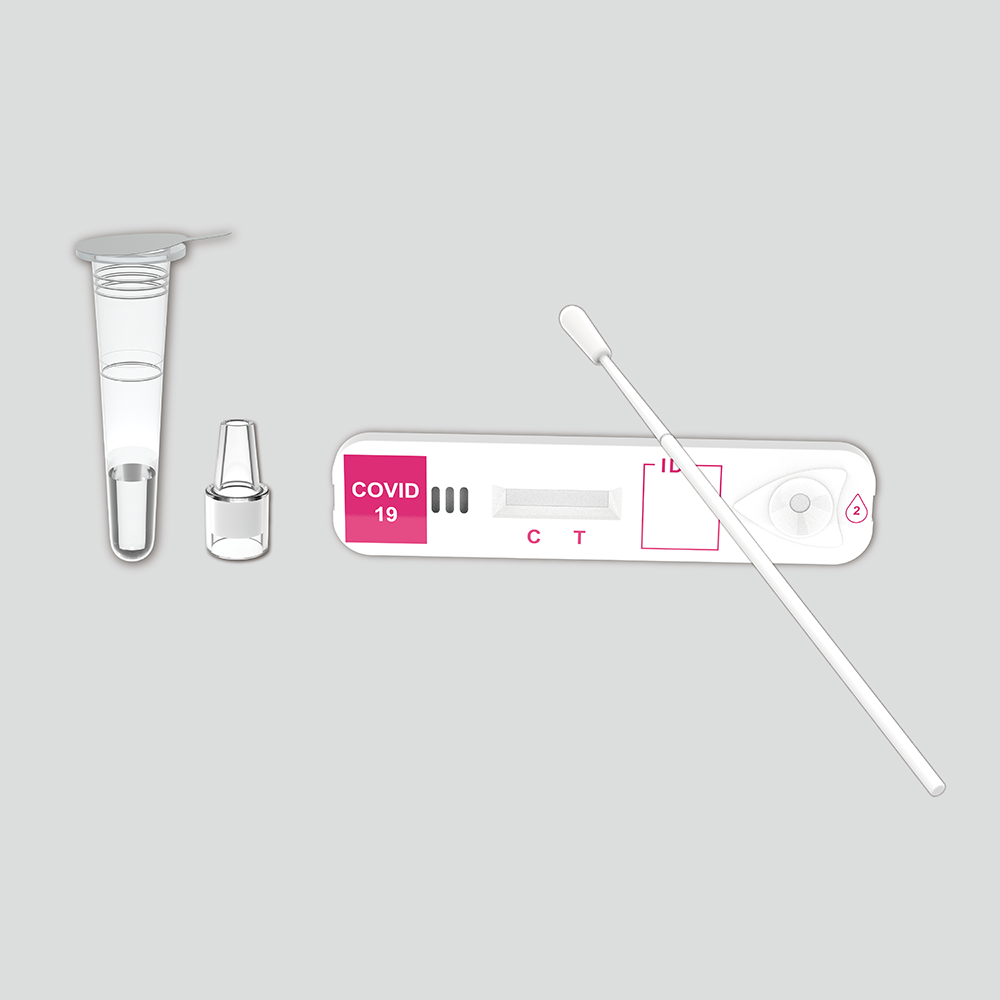
KaiBiLiTM Flu & COVID-19 Antigen Duo Rapid Test is intended for the simultaneous qualitative detection and differentiation of the nucleocapsid protein antigens from SARS-CoV-2, influenza A and influenza B in direct nasopharyngeal (NP) specimens. The detection is based on the antibodies which were developed specifically recognizing and reacting with the nucleoprotein of virus.
Detection
KaiBiLiTM Flu & COVID-19 Antigen Duo Rapid Test is the simultaneous qualitative detection and differentiation of the nucleocapsid protein antigens from SARS-CoV-2, influenza A and influenza B in direct nasopharyngeal (NP) specimens.
Specimen
Nasopharyngeal
Limit of Detection (LoD)
SARS-CoV-2 & Flu: 140 TCID50/mL
Influenza A
|
Influenza Viral Strain |
Calculated LOD (TCID50/mL) |
| A/New Caledonia/20/1999_H1N1 |
8.50×103 |
| A/California/04/2009_H1N1 |
2.11×103 |
| A/PR/8/34_H1N1 |
2.93×103 |
| A/Bean Goose/Hubei/chenhu XVI35-1/2016_H3N2 |
4.94×102 |
| A/Guizhou/54/89_H3N2 |
3.95×102 |
| A/Human/Hubei/3/2005_H3N2 |
2.93×104 |
| A/Bar-headed Goose/QH/BTY2/2015_H5N1 |
1.98×105 |
| A/Anhui/1/2013_H7N9 |
7.90×105 |
Influenza B.
|
Influenza Viral Strain |
Calculated LOD (TCID50/mL) |
| B/Victoria |
4.25×103 |
| B/Yamagata |
1.58×102 |
Accuracy
| Influenza A | Influenza B | COVID-19 | |
| Relative Sensitivity | 86.80% | 91.70% | 96.60% |
| Relative Specificity | 94% | 97.50% | 100% |
| Accuracy | 92.20% | 96.10% | 98.90% |
Time to Results
Read results at 15minutes and no more than 30 minutes.
Kit storage conditions
2~30°C.
Contents
| Test devices | 20 tests |
| Sterilized swabs | 20 pcs. |
| Extraction tubes (with 0.5mL extraction buffer) | 20 pcs. |
| Nozzle with filter | 20 pcs. |
| Tube Stand | 1 pcs. |
| Package Insert | 1 pcs. |
Ordering Information
|
Product |
Cat.No. |
Contents |
|
KaiBiLiTM Flu & COVID-19 Antigen Duo |
P211137 |
20 Tests |
Product detail pictures:

Related Product Guide:
We emphasize advancement and introduce new products into the market each year for China Gold Supplier for Antigen Diagnostic Test - KaiBiLi Flu&Covid-19 Antigen Duo Rapid Test – Genesis , The product will supply to all over the world, such as: Argentina, Porto, Germany, During in 11 years, We have participated in more than 20 exhibitions, obtains the highest praise from each customer. Our company has been devoting that "customer first" and committed to helping customers expand their business, so that they become the Big Boss !
This supplier stick to the principle of "Quality first, Honesty as base", it is absolutely to be trust.